Surfactant Fundamentals
Pierre-Gilles de Gennes , 1991- Physics Nobel Price, says:
"Without Surfactants we would be powerless confronted to 90% of the Industrial Problems".
Basic Concepts
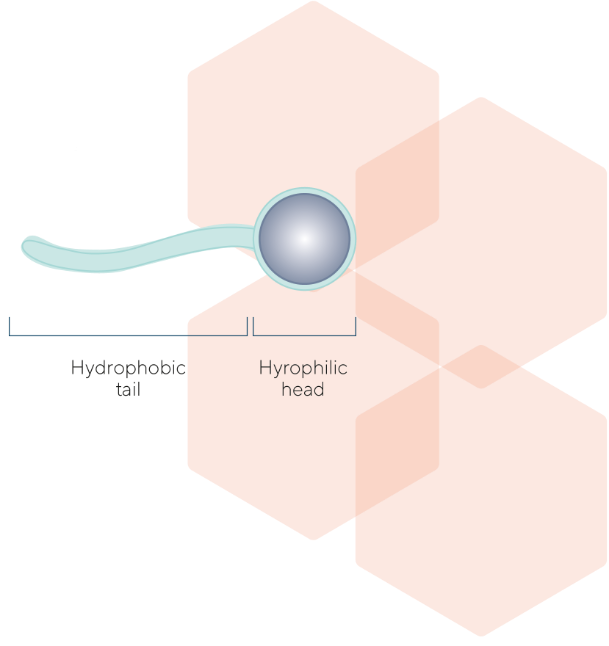
The impressive number of useful applicative effects that surfactants may deliver derived from their particular chemical structure: a non-polar hydrophobic moiety linked through a chemical bond to a polar hydrophilic moiety.
Interfase adsorbtion
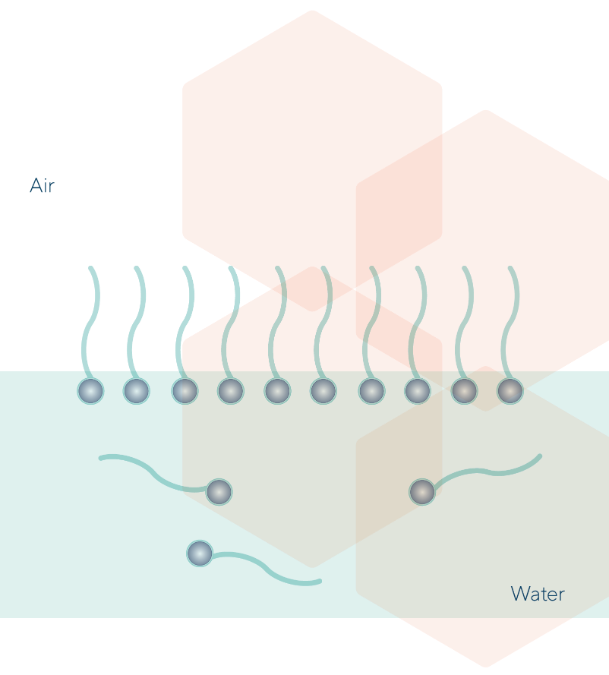
Surfactants dissolved in water migrate to the air-water interface with their hydrophobic tails oriented toward the air and the hydrophilic heads towards the water. That causes the water Surface Tension reduction
Micelles Formation
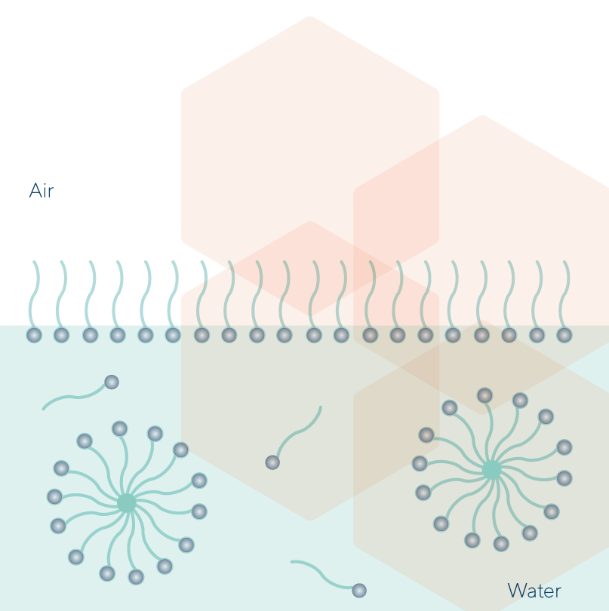
When the surfactant monolayer has completely covered the water solution surface, further dissolved surfactant molecules will aggregate into micelles (Critical Micelles Concentration: CMC)
Different types of Micelles
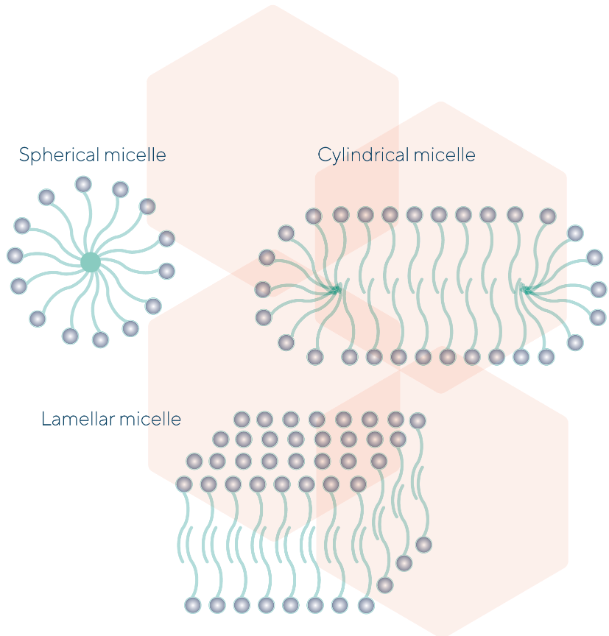
At the higher surfactant concentration the micelles may appear in different shapes, for examples cylindrical and lamellar
Performace properties
Surfactants are able to change the properties of the interfaces (through the interface adsorption) and the solution phase (through the micellization): these two phenomena rules all the useful practical effects that surfactants can deliver
Performance Properties derived from the Interface Adsorbtion
Wetting:
Water does not spread on hydrophobic substrates like waxy or oily surfaces. The addition of a surfactant leads to the interface adsorption. The water/substrate interfacial tension will decrease and the tendency of the surfactant solution to wet the substrate will increase.
Surfactants with relatively low molecular weight and with branched alkyl chains are applied as wetting agents.
Waterproofing:
When want to render a polar substrate more hydrophobic, the addition of relevant surfactant causes the interfacial adsorption,with the hydrophobic moiety oriented toward the water and the hydrophilic moiety oriented toward the substrate. The water/substrate interfacial tension will increase and the tendency of the surfactant solution to wet the substrate will decrease.
Owing to that polar substrates are very often negatively charged, surfactants for waterproofing are normally cationic surfactants that have strong adsorption properties with their positively charged head on the negatively charged substrate.
This effect is the basis of wool softening treatments and of the hair conditioning, therefore both textile softeners and hair conditioner formulations contain cationic surfactants.
Emulsion is a dispersion (droplets) of a liquid in another immiscible liquid: the phase that is present in the form of droplets is called as dispersed or internal phase, and the phase in which the droplets are suspended as continuous or external phase.
One of the liquids is quite often water and the other one is an “oil” (nearly insoluble organic liquid). If the oil phase is dispersed in the aqueous phase, we have an “oil in water” (O/W) emulsion, or “water in oil” (W/O) emulsion in the opposite case. Water and immiscible “oil” liquids do not form spontaneously an emulsion.
Adding a surfactant leads to the interface adsorption: the water/oil interfacial tension will decrease. If mechanical energy in the form of a mixer is added to the system, it might be possible to form an emulsion.
However, it is not sufficient to give the resulting emulsion of a significant stability. In order to reach this target, the surfactant adsorption process must produce sufficient high energy barriers surrounding the discontinuous phase particles to prevent their re-aggregation. This stabilization may be electrostatic or steric in nature.
The electrostatic stabilization is generated by the adsorption onto the particles of ionic, mainly anionic, surfactants in order to give each particle a charge of the sign similar to that of the adsorbed surfactant. As a consequence the particles will repel each other.
The steric stabilization is in general generated by the adsorption onto the particles of nonionic surfactants with hydrophilic moiety oriented toward the water and having sufficient bulk to constitute an efficient barrier to the close approach of two or more particles. Typically, the nonionic surfactant is a polyoxyethylene derivative. The efficient bulk barrier is given by the long polyoxyethylene chain, extending into the aqueous phase and surrounding the discontinuous phase particles.
Demulsification of an existing emulsion occurs when electrostatic or steric barriers are reduced or eliminated resulting in a “breaking” of the emulsion.
The same concepts as for emulsification and demulsification apply when the discontinuous phase is a solid.
Dispersion of solid particles in liquid, where they are insoluble, is stabilized by the interface adsorption of the surfactant molecules that produce electrostatic or steric barriers to the re-aggregation. In this case the mechanical energy is added by a mill.
Ethoxylated anionic surfactants, that can combine electrostatic and steric stabilization, have good dispersion properties.
Surfactants with hydrophobic moieties containing aromatic rings are able to strongly adsorb onto the solid so they are suitable to disperse solid particles in water, for example the anionic tristyrylphenol derivatives.
Polymeric surfactants as well show excellent dispersion performances: our hyperdispersing agents are able to offer outstanding dispersing power for both water and solvent systems.
On the contrary the addition of surfactants which favorites the reduction or elimination of the electrostatic or steric barriers cause flocculation.
Foam is the dispersion of a gas in a liquid produced when the gas is introduced into a solution whose surface film has viscoelastic properties.
Pure water does not foam, but if we add a surfactant with the proper structure the interface adsorption will imparts viscoelatic properties to the interfacial film and the solution will foam. Surfactants with the ability to form a closely packed layer at the air/water interface,improving in this way the stability of the foam, are considered as very good foaming agents.
Generally anionic surfactants are better foaming than nonionic.
In defoaming the added surfactant eliminates or reduces the viscoelastic properties of the interfacial film causing the foam collapse. Surfactants that adsorb at the interface to form a loosely packed film, are particularly effective as defoaming agents , for example those with highly branched alkyl chain or with polyoxyethylene chain ended (“capped”) with a polyoxypropylene chain.
Performance Properties derived from Micelles Formation
The presence of micelles in a solution makes possible the solubilization of liquid or solid materials that are normally insoluble in the solvent.
In water solutions the polar materials are usually solubilized in the outer region of the micelle close to the hydrophilic head groups, whereas non polar materials are solubilized in the inner core of the micelle between the hydrophobic groups.
Aqueous solutions of spherical micelles have very low viscosities. More complex micelles arrangements like the liquid crystals give high viscosities.
Combined with proper surfactants cylindrical micelles or liquid crystals at relatively low surfactant concentration are obtainable. This effect is important in formulations of personal care products, like shampoos, shower gels or foam baths, where the total surfactant concentration has to be maintained at relatively low values and at the same time relatively high viscosities are needed.
The presence of liquid crystals in surfactant solutions is often undesirable since the associated high viscosity can limit the solubility of other solutes in the solution. Liquid crystals formation can be inhibited by the use of surfactant materials known as hydrotropes that are able to disrupt the parallel alignments of the liquid crystal structures.
Performance Properties derived from both Interface Adsorbtion and Micellization
Detergency is a complex process that involves both interface adsorption (wetting, emulsification and dispersion of the dirt particles) and micellization (solubilization of the dirt particles into the micelles).
